DIN EN ISO 9001:2015
Quality: we work according to our customers’ standards
Our priority is to ensure quality, functionality, and safety in our products. We set the highest standards in everything. We adhere strictly to internal and external requirements in product development, processing, and handling.
Aero Pump is known for high-quality products that fulfil the agreed specification across our full product range and in new developments.
This has created the confidence that our customers place in us, confidence we have earned by consistently striving to manufacture and supply products in excellent quality.
Quality assurance
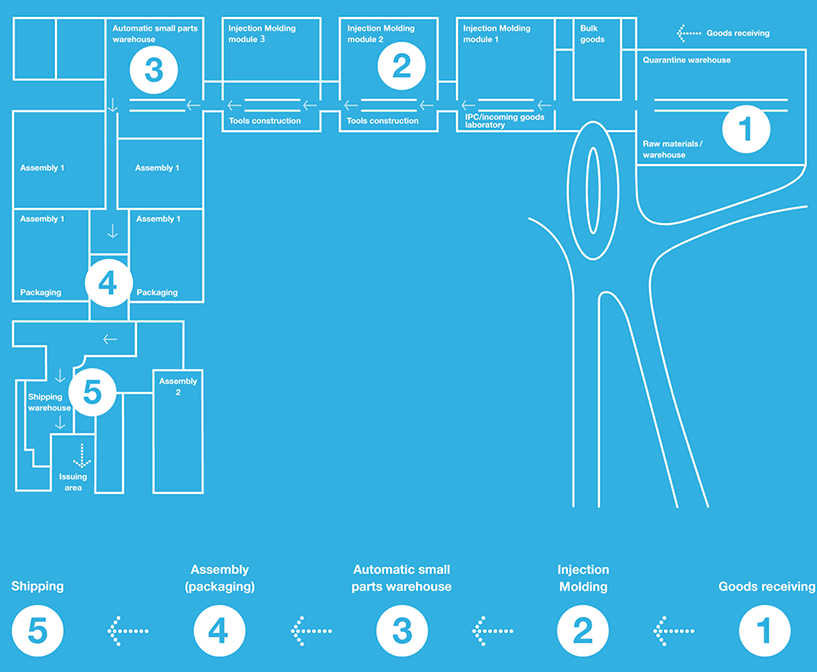
The Aero Pump manufacturing building’s modular interior design follows the value-added chain in compliance with the demanding guidelines of GMP regulations for the pharmaceutical sector. Our testing areas are equipped with the corresponding test equipment to ensure reliable quality assessment every step of the way. Incoming goods inspection, in-process controls, and final inspection are a matter of routine. Consistently separating these areas from production reflects our priorities on quality assessment.
Training management
These requirements mean that we need to develop constantly as a company. Activities change and new areas of responsibility emerge. Demand for qualified staff plays a central role in the pharmaceutical regulations, so we have taken this as a model and created an internal organisation to be able to offer training and qualification programmes for our employees. Our concept of quality centres on securing, maintaining, and documenting our expertise.
The following resources provide support in our everyday work:
- Integrated management in quality, environment, hygiene, and relevant GMP requirements
- Internal training management
- Automation in our logistics, injection moulding, and assembly processes
- Close cooperation and direct communication with customers and suppliers as well as amongst colleagues minimise the risk of error
- State-of-the-art technology from goods receipt to materials testing, injection moulding, assembly, and shipping
- State-of-the-art safety concepts in our production flow
- Production flexibility from design to production makes it possible to increase capacity fast and expand on our product range in the short term
Certifications
Aero Pump is certified according to DIN EN ISO 9001 (quality management system), DIN EN ISO 14001 (environmental management system), DIN EN ISO 50001 (energy management system), DIN EN ISO 15378 (primary packaging for medicinal products) and DIN EN ISO 13485 (medical devices). We combine these management systems to meet all the requirements of our stakeholders.
Certified quality management
Material flow
Efficiency begins with architecture
Our new Aero Pump headquarters building in Hochheim corresponds to modern workflows with specific focus on the requirements the pharmaceutical industry places on production. These requirements included cleanroom production in injection moulding and assembly, separated storage areas, labelling, and documentation in production.